Research Insight
Study on the Antitumor Effects and Molecular Mechanisms of Sanghuangporus Polysaccharides 
2 College of Pharmacy, Changchun University of Chinese Medicine, Changchun, 130117, Heilongjiang, China;
3 Institute of Antler Science and Product Technology, Changchun Sci-Tech University, Changchun, 130012, Heilongjiang, China;
4 School of Food Science and Technology, Jiangnan University, Wuxi, 214122, Jiangsu, China
 Author
Author  Correspondence author
Correspondence author
International Journal of Molecular Medical Science, 2024, Vol. 14, No. 6
Received: 14 Sep., 2024 Accepted: 22 Oct., 2024 Published: 06 Nov., 2024
This study explores the antitumor effects and molecular mechanisms of Sanghuangporus polysaccharides. As the main active components of the traditional medicinal mushroom, Sanghuangporus polysaccharides exhibit significant antioxidant, immunomodulatory, and antitumor activities. They inhibit tumor growth by regulating the cell cycle, promoting apoptosis, and reducing cancer cell migration and invasion. Sanghuangporus polysaccharides exert antitumor effects by activating the p53 signaling pathway and down-regulating the expression of matrix metalloproteinases (MMPs). Additionally, their antioxidant properties help alleviate the side effects of chemotherapy and radiation therapy in cancer patients. Sanghuangporus polysaccharides can also be used for cancer prevention and adjuvant therapy, making them a promising natural therapeutic agent.
1 Introduction
Sanghuangporus, a traditional medicinal mushroom, has been utilized in Asia for nearly 2 000 years due to its various health benefits, including its potential anticancer properties (Wan et al., 2020; Wang et al., 2022b). Polysaccharides derived from Sanghuangporus have garnered significant attention for their biological activities, such as antioxidant, anti-aging, immunomodulatory, and particularly antitumor effects (Wang et al., 2022b; Zhou et al., 2023). Recent studies have demonstrated that these polysaccharides can inhibit tumor growth and induce apoptosis in cancer cells, although the precise molecular mechanisms remain to be fully elucidated (Chen and Huang, 2018; Wan et al., 2020). The increasing incidence of cancer and the severe side effects associated with conventional chemotherapy drugs underscore the urgent need for alternative therapeutic agents with fewer adverse effects (Xie et al., 2020).
Polysaccharides derived from Phellinus linteus exhibit a wide range of biological activities, with antitumor properties being particularly noteworthy. These polysaccharides have been shown to inhibit tumor growth and proliferation in various cancer cell lines. For instance, Sanghuangporus vaninii polysaccharides (SVP) have demonstrated modest anti-proliferative activity against HeLa, SHG-44, SMMC-7721, and MCF-7 cells. They effectively regulate the cell cycle, promote apoptosis, and reduce the migratory and invasive capacities of cancer cells. The antitumor mechanisms of these polysaccharides involve the activation of p53-related genes and the down-regulation of matrix metalloproteinase (MMP) expression, which are crucial for tumor suppression (Wan et al., 2020). Additionally, polysaccharides from Sanghuangporus Sanghuang have been found to possess antioxidant, anti-inflammatory, and antibacterial properties, further contributing to their therapeutic potential (Zhou et al., 2023; Liu et al., 2023b).
This study aims to provide a comprehensive overview of the antitumor effects and molecular mechanisms of Sanghuangporus polysaccharides, discuses the traditional applications and medicinal value of Phellinus linteus, as well as the specific biological activities of its polysaccharides, seeks to elucidate the potential of these natural compounds in cancer therapy. This study reviews an analysis of the extraction, purification, and structural characterization of these polysaccharides, as well as their specific antitumor mechanisms and broader biological activities. Through this study detailed examination, hopes to highlight the therapeutic potential of Sanghuangporus polysaccharides and their possible applications in modern medicine.
2 Basic Properties and Chemical Composition of Sanghuangporus Polysaccharides
2.1 Extraction and isolation of Sanghuangporus polysaccharides
The extraction, isolation, and purification of polysaccharides form the basis for further research into their monosaccharide composition, molecular weight, primary structure, types and degrees of branching, configuration, biological activity, and structure-activity relationship. Sanghuangporus polysaccharides can be extracted using various methods, including hot water extraction and ultrasonic-assisted extraction. For instance, the optimal conditions for ultrasonic-assisted extraction from Sanghuangporus vaninii were found to be an extraction temperature of 60°C, an extraction time of 60 minutes, a solid-liquid ratio of 40 g/mL, and an ultrasonic power of 70 W, achieving an extraction rate of 1.41% (Liu et al., 2023b). Another method involves the use of a thermoseparating aqueous two-phase system (ATPS) based on ethylene oxide-propylene oxide (EOPO) copolymer and deep eutectic solvents (DES), which has shown high efficiency in extracting polysaccharides (Figure 1; Figure 2) (Gao et al., 2020; Wang et al., 2022a).
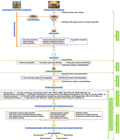 Figure 1 Schematic illustration of preparation, structural elucidation, and bioactivity determination of the polysaccharides derived from the naturallymedicinal fungal resources Sanghuangporus (Adopted from Wang et al., 2022a) |
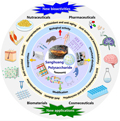 Figure 2 The overall highlights discussed in this review (Adopted from Wang et al., 2022a) |
Purification and isolation of Sanghuangporus polysaccharides often involve gradient ethanol precipitation and chromatographic techniques. For example, five homogeneous monosaccharides were obtained by gradient ethanol precipitation followed by diethylaminoethyl-cellulose (DEAE) and Sephadex G-100 separation and purification (Liu et al., 2023b). High-speed counter-current chromatography (CCC) using an aqueous diphase solvent system has also been successfully employed for one-step separation of polysaccharides, minimizing chemical variation (Yin et al., 2012).
The chemical structure of Sanghuangporus polysaccharides can be preliminarily identified using various analytical techniques. High-performance liquid chromatography (HPLC), ultraviolet (UV) spectroscopy, Fourier transform infrared (FT-IR) spectroscopy, thermogravimetric analysis (TGA), Zeta potential analysis, and scanning electron microscopy (SEM) are commonly used to characterize the polysaccharides (Liu et al., 2023b). These techniques help in determining the monosaccharide composition, glycosidic linkages, and molecular weight distribution.
2.2 Chemical composition analysis of Sanghuangporus polysaccharides
Sanghuangporus polysaccharides are composed of various monosaccharides, including glucose, galactose, mannose, xylose, arabinose, and rhamnose. These components contribute to the biological activities of the polysaccharides, such as antioxidant and anti-inflammatory effects (Liu et al., 2018; Liu et al., 2023b). The specific composition and structure can vary depending on the extraction and purification methods used (Table 1).
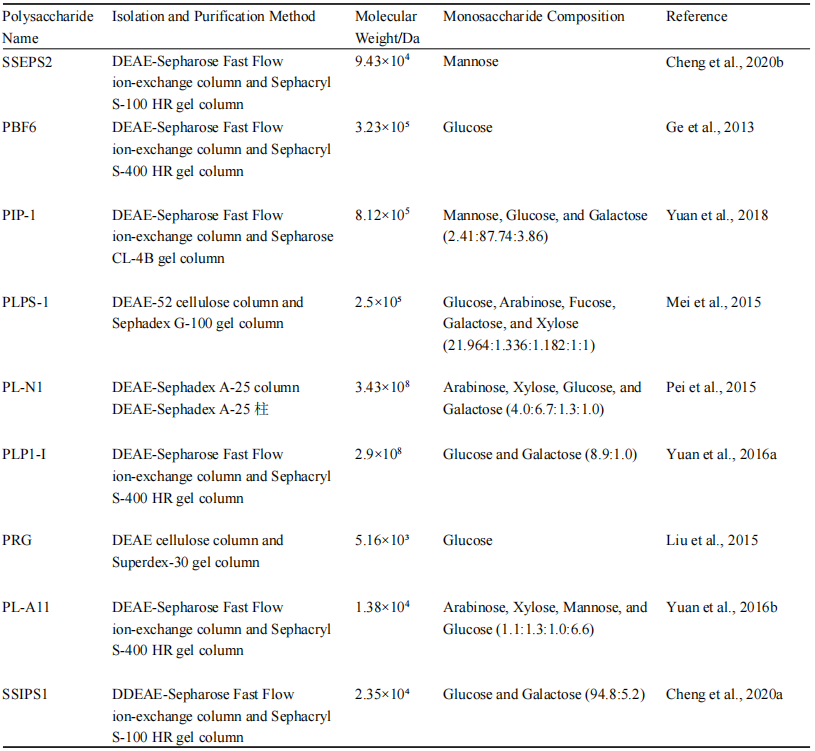 Table 1 Structural analysis of Sanghuangporus polysaccharides (Li et al., 2022) |
The molecular weight of Sanghuangporus polysaccharides can vary significantly, and their structural complexity is revealed through techniques such as viscometry, osmometry, precipitation methods, HPLC, GPC, HPGPC, HPSEC, SEC-MALLS-RI, and HPSEC-MALLS-RI. For example, polysaccharides purified from Sanghuangporus fungi have an average molecular weight of 20.377 kDa (Zhang et al., 2023). Structural analysis indicates that the molecular weight of these polysaccharides ranges from 1 to 1×10⁶ kDa, and there is a correlation between molecular weight and the antioxidant capacity of the polysaccharides. Low-molecular-weight polysaccharides exhibit significantly better antioxidant and free radical scavenging abilities than high-molecular-weight polysaccharides, possibly due to the more active intramolecular hydrogen bonding of O-H and electron-donating substituents. Structural analysis using methylation and two-dimensional nuclear magnetic resonance (2D-NMR) techniques indicates that these polysaccharides typically consist of complex glycosidic linkages, including 1,4-linked α-D-pyran glucose residues with branching points (Figure 3) (Cheng et al., 2020a).
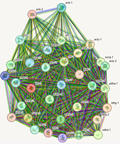 Figure 3 Molecular diagram |
Sanghuangporus polysaccharides have shown potential antitumor effects. They can inhibit the levels of pro-inflammatory cytokines and promote anti-inflammatory cytokines, which may contribute to their antitumor activity (Liu et al., 2023b). Additionally, these polysaccharides have been found to downregulate genes involved in lipid accumulation, suggesting a role in lipid regulation therapy (Zhang et al., 2023).
2.3 Biological activities of Sanghuangporus polysaccharides
Sanghuangporus polysaccharides exhibit significant antioxidant activity. They have been shown to scavenge superoxide anions and other free radicals, which helps in reducing oxidative stress (Liu et al., 2018; Liu et al., 2023b). The antioxidant properties are influenced by the specific monosaccharide composition and molecular weight of the polysaccharides.
The immunomodulatory effects of Sanghuangporus polysaccharides include enhancing macrophage activity and modulating cytokine production. For example, certain fractions of Lycium barbarum polysaccharides, which share similar properties with Sanghuangporus polysaccharides, have been shown to enhance macrophage NO production and phagocytic capacity. These effects contribute to the overall immune-boosting properties of the polysaccharides.
Preliminary studies have shown that Sanghuangporus polysaccharides possess antitumor potential. They can inhibit the proliferation of tumor cells and modulate immune responses to more effectively target cancer cells (Liu et al., 2023a; Zhang et al., 2023). Wei et al. (2016) studied the inhibitory effect of Sanghuangporus polysaccharides on liver cancer and found that the tumor mass in the experimental group treated with Sanghuangporus polysaccharides was reduced, and liver cancer cell necrosis was observed. Compared to anticancer drugs, Sanghuangporus polysaccharides demonstrated lower toxicity. Analysis of the antitumor mechanism suggested that Sanghuangporus polysaccharides might be related to immune regulation. Ying et al. (2017) found through research that polysaccharides from Sanghuangporus fruiting bodies exhibited better antitumor effects than those from Sanghuangporus mycelium. Additionally, the antitumor activity of mycelium polysaccharides gradually increased as the growth period extended.
However, further research is still needed to fully understand the mechanisms behind these antitumor effects and to develop potential therapeutic applications. Sanghuangporus polysaccharides exhibit a range of biological activities, including antioxidant, immunomodulatory, and antitumor effects. Their chemical composition and structural characteristics play a crucial role in determining these activities, making them a promising area for research in therapeutic applications.
3 Inhibitory Effects of Sanghuangporus Polysaccharides on Tumor Cell Proliferation
3.1 In vitro studies
Sanghuangporus polysaccharides have garnered significant attention for their antitumor potential in various in vitro studies. These polysaccharides can inhibit tumor cell proliferation and modulate immune responses to target cancer cells more effectively. For instance, polysaccharides isolated from Sanghuangporus vaninii (SVP) exhibited moderate antiproliferative activity against several cancer cell lines, including HeLa, SHG-44, SMMC-7721, and MCF-7 cells. In tumor cell inhibition experiments conducted in vitro, both crude polysaccharides and the purified SSP1 were shown to inhibit the proliferation of tumor cells such as HepG2, A549, MDA-MB-231, PC3, SGC-7901, and HCT-116 to varying degrees. Notably, SSP1 was more effective than the crude polysaccharides, with a significant difference (P<0.05) (Ying, 2023). Additionally, research has shown that SVP can modulate the cell cycle, promote apoptosis, and reduce the migration and invasion ability of MCF-7 cells (Figure 4) (Wan et al., 2020).
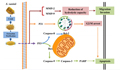 Figure 4 A series of antitumor mechanism pathways for SVP in MCF-7 cells (Adopted from Wan et al., 2020) |
Polysaccharides extracted from Polygala tenuifolia Willd. (PTP) were found to induce apoptosis in S180 sarcoma cells, characterized by nuclear fragmentation, apoptotic body formation, and a decrease in mitochondrial membrane potential (Yu et al., 2021a). Cheng et al. (2020a) isolated a novel extracellular polysaccharide from the liquid culture of Sanghuangporus species, composed solely of mannose (Man), which featured 1,3-linked and 1,2-linked α-D-Manp units. The 1,2-linked α-D-Manp was substituted at the O-6 position with 1,6-linked α-D-Manp residues and terminal α-D-Manp residues, demonstrating potential antitumor activity against HepG2 and MCF-7 cell growth in vitro.
Ganoderma atrum polysaccharide (PSG-1) also showed antitumor effects by inhibiting tumor growth and inducing apoptosis in tumor cells. The treatment with PSG-1 resulted in increased cAMP and PKA activities, which are crucial for the apoptotic pathway (Zhang et al., 2014a). Similarly, polysaccharides from Lentinus edodes (SLNT1 and JLNT1) demonstrated significant antitumor effects by inducing apoptosis in H22-bearing mice (Wang et al., 2013).
3.2 Molecular mechanisms of cell cycle regulation
The molecular mechanisms underlying the antitumor effects of Sanghuangporus polysaccharides involve several pathways. SVP from Sanghuangporus vaninii was found to enhance the activation of p53-related genes and down-regulate matrix metalloproteinase (MMP) expression, which are critical for cell cycle regulation and apoptosis (Wan et al., 2020). The PTP from Polygala tenuifolia Willd. induced apoptosis through the FAS/FAS-L-mediated death receptor pathway, increasing the ratio of BAX to Bcl-2, promoting cytochrome c release, and activating caspase-9 and -3 (Yu et al., 2021a).
Ganoderma atrum polysaccharide (PSG-1) activated the cAMP-PKA signaling pathway while down-regulating the Ca(2+)/PKC signal pathway, leading to apoptosis in tumor cells (Zhang et al., 2014a). Another study on PSG-1 revealed that it induced apoptosis via the mitochondria-mediated pathway, involving the loss of mitochondrial membrane potential, cytochrome c release, and activation of caspases (Zhang et al., 2014b). Furthermore, polysaccharides from Tetrastigma Hemsleyanum (SYQ-PA) were shown to inhibit breast cancer by reprogramming tumor-associated macrophages through the PPARγ signaling pathway, promoting M1 macrophage polarization and inhibiting cancer cell proliferation (Liu et al., 2023a).
3.3 In vivo experimental studies
In vivo studies have further confirmed the antitumor efficacy of Sanghuangporus polysaccharides. For example, SVP from Sanghuangporus vaninii significantly inhibited tumor growth in MCF-7 xenograft models by promoting apoptosis and reducing tumor invasiveness (Wan et al., 2020). Polysaccharides from Polygala tenuifolia Willd. (PTP) demonstrated antitumor activity in S180 tumor-bearing mice by inducing apoptosis and enhancing immune responses, such as increased NK cell activity and cytokine production (Yu et al., 2021a).
Ganoderma atrum polysaccharide (PSG-1) showed potent antitumor effects in CT26 tumor-bearing mice by inducing apoptosis and boosting the immune system, as evidenced by increased lymphocyte proliferation and cytokine levels (Zhang et al., 2014b). Similarly, polysaccharides from Lentinus edodes (SLNT1 and JLNT1) significantly inhibited tumor growth in H22-bearing mice by enhancing immune responses and inducing apoptosis (Wang et al., 2013).
Polysaccharides from Actinidia eriantha roots (AEP) also exhibited significant antitumor activity in mouse models by promoting splenocyte proliferation, NK cell activity, and cytokine production, thereby enhancing the immune response (Xu et al., 2009). Additionally, polysaccharides from Panax japonicus (PSPJ) repressed H22 tumor growth in vivo by modulating immune responses and reducing immunosuppressive factors in tumor-associated macrophages (Shu et al., 2018).
Sanghuangporus polysaccharides exhibit potent antitumor effects through various mechanisms, including cell cycle regulation, apoptosis induction, and immune modulation. These findings highlight the potential of Sanghuangporus polysaccharides as natural therapeutic agents for cancer treatment.
4 Mechanisms of Sanghuangporus Polysaccharides-Induced Tumor Cell Apoptosis
4.1 Methods for detecting apoptosis
To investigate the antitumor effects of Sanghuangporus polysaccharides, various methods are employed to detect apoptosis in cancer cells. Common techniques include the Cell Counting Kit-8 (CCK-8) assay, which measures cell viability, and Hoechst staining, which identifies apoptotic cells by their characteristic nuclear morphology (Figure 5) (Ding et al., 2020). Flow cytometry is another critical method used to analyze cell cycle distribution and quantify apoptotic cells by detecting sub-G1 peaks (Cao et al., 2010). Additionally, TUNEL assay and PI staining are utilized to confirm DNA fragmentation and other morphological changes associated with apoptosis (Song et al., 2014; Chen, 2024).
 Figure 5 Inhibitory effects of Sargassum fusiforme polysaccharide (SFPS) on the growth of human erythroleukemia (HEL) cells and human embryonic lung (MRC-5) cells relative to DMEM-treated control cultures (Adopted from Ding et al., 2020) Image caption: Data are presented as the mean ± SD (n=5). HEL cells and MRC-5 cells were incubated with (A) SFPS I, (B) SFPS II, and (C) SFPS III (10~100 μg·mL−1) for 24 h. (D) IC50 values for the inhibition of HEL cell viability by different purified components of SFPS. Cell viability was measured using CCK-8 and MTT assays (Adopted from Ding et al., 2020) |
Western blot analysis is frequently used to detect the expression of apoptosis-related proteins, providing insights into the molecular mechanisms underlying apoptosis (Cao et al., 2010; Bie et al., 2020). Real-time quantitative polymerase chain reaction (qPCR) is employed to measure the mRNA levels of genes involved in apoptosis, offering a comprehensive understanding of gene regulation during the apoptotic process (Ding et al., 2020; Xu et al., 2021).
4.2 Study of apoptotic signaling pathways
Sanghuangporus polysaccharides induce apoptosis through multiple signaling pathways. One of the primary pathways is the intrinsic mitochondrial pathway, which involves the disruption of mitochondrial membrane potential and the release of cytochrome c into the cytoplasm. This pathway is characterized by the activation of caspase-9 and caspase-3, leading to the execution of apoptosis (Cao et al., 2010; Wu et al., 2014). The intrinsic pathway is often regulated by the balance between pro-apoptotic (e.g., Bax) and anti-apoptotic (e.g., Bcl-2) proteins (Figure 6) (Cui et al., 2016; Wan et al., 2020).
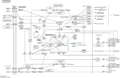 Figure 6 Mechanism diagram of cell apoptosis signaling pathway |
Another significant pathway is the extrinsic death receptor pathway, which involves the binding of ligands to death receptors such as FAS, leading to the activation of caspase-8 and subsequent activation of downstream caspases (Yu et al., 2020). This pathway can also interact with the intrinsic pathway through the cleavage of Bid, a pro-apoptotic Bcl-2 family member, linking the two apoptotic mechanisms (Carneiro and El-Deiry, 2020).
Additionally, Sanghuangporus polysaccharides have been shown to modulate other signaling pathways, such as the PI3K/AKT and MAPK pathways. These pathways play crucial roles in cell survival and proliferation, and their inhibition can promote apoptosis. For instance, the polysaccharide from Grifola frondosa (GFP-A) was found to inhibit the PI3K/AKT pathway and activate the MAPK pathway, leading to increased apoptosis in HT-29 cells (Bie et al., 2020). Similarly, the AMPK/PI3K/mTOR pathway is involved in the regulation of apoptosis and autophagy, with polysaccharides from Pleurotus nebrodensis (PN50G) shown to activate AMPK and inhibit PI3K/AKT, promoting apoptosis in A549 cells (Cui et al., 2016).
4.3 Expression of apoptosis-related genes and proteins
The expression of apoptosis-related genes and proteins is a critical aspect of understanding the molecular mechanisms of Sanghuangporus polysaccharides-induced apoptosis. Key genes involved in the intrinsic pathway include Bax, Bcl-2, and caspases. Studies have shown that treatment with Sanghuangporus polysaccharides leads to the upregulation of Bax and downregulation of Bcl-2, promoting the release of cytochrome c and activation of caspase-9 and caspase-3 (Wu et al., 2014; Wan et al., 2020). This regulation of gene expression is crucial for the initiation and execution of apoptosis.
In the extrinsic pathway, the expression of death receptors such as FAS and their ligands (FAS-L) is upregulated, leading to the activation of caspase-8 and subsequent apoptosis (Yu et al., 2020). The interaction between the intrinsic and extrinsic pathways is further evidenced by the cleavage of Bid, linking the two apoptotic mechanisms (Song et al., 2014).
Moreover, the modulation of other signaling pathways also affects the expression of apoptosis-related genes and proteins. For example, the inhibition of the PI3K/AKT pathway by GFP-A results in decreased expression of anti-apoptotic proteins and increased expression of pro-apoptotic proteins, enhancing apoptosis in HT-29 cells (Bie et al., 2020). Similarly, the activation of AMPK and inhibition of mTOR by PN50G leads to changes in the expression of proteins involved in cell cycle regulation and apoptosis, promoting cell death in A549 cells (Cui et al., 2016).
Sanghuangporus polysaccharides induce tumor cell apoptosis through multiple mechanisms, involving the intrinsic and extrinsic pathways, as well as the modulation of other signaling pathways. The detection of apoptosis, study of signaling pathways, and analysis of gene and protein expression provide a comprehensive understanding of the molecular mechanisms underlying the antitumor effects of Sanghuangporus polysaccharides. These findings highlight the potential of Sanghuangporus polysaccharides as promising agents for cancer therapy.
5 Antioxidant Stress Effects of Sanghuangporus Polysaccharides and Their Antitumor Mechanisms
5.1 Relationship between oxidative stress and tumor development
Oxidative stress, characterized by an imbalance between the production of reactive oxygen species (ROS) and the body's ability to detoxify these reactive intermediates, plays a crucial role in the development and progression of tumors. Excessive ROS can damage cellular components, including DNA, proteins, and lipids, leading to mutations and genomic instability, which are hallmarks of cancer. The persistent oxidative environment promotes oncogenic signaling pathways, such as those involving NF-κB and HIF, which further drive tumorigenesis by enhancing cell proliferation, survival, and angiogenesis (Jiao et al., 2016).
Moreover, oxidative stress can modulate the tumor microenvironment, making it more conducive to cancer progression. For instance, ROS can activate stromal cells and immune cells within the tumor microenvironment, leading to the secretion of pro-inflammatory cytokines and growth factors that support tumor growth and metastasis (Jiao et al., 2016). Therefore, targeting oxidative stress and its associated pathways presents a promising strategy for cancer prevention and therapy.
5.2 Analysis of antioxidant activity of Sanghuangporus polysaccharides
Sanghuangporus polysaccharides have demonstrated significant antioxidant activities, which are pivotal in their antitumor effects. These polysaccharides can scavenge various free radicals, including DPPH, superoxide, and hydroxyl radicals, thereby reducing oxidative stress within the body (Shao et al., 2014; Wang et al., 2018). The antioxidant potential of Sanghuangporus polysaccharides is dose-dependent, with higher concentrations exhibiting greater free radical scavenging abilities (Wang et al., 2018).
In addition to direct free radical scavenging, Sanghuangporus polysaccharides can enhance the body's endogenous antioxidant defense systems. For example, they have been shown to upregulate the expression of antioxidant enzymes such as superoxide dismutase (SOD) and catalase, which play critical roles in neutralizing ROS (Wang et al., 2018). This dual mechanism of action-direct scavenging of free radicals and boosting endogenous antioxidant defenses-makes Sanghuangporus polysaccharides potent agents against oxidative stress.
5.3 Mechanisms of tumor inhibition by Sanghuangporus polysaccharides through antioxidant stress
Sanghuangporus polysaccharides inhibit tumor growth through multiple mechanisms, primarily by mitigating oxidative stress. By reducing ROS levels, these polysaccharides prevent the oxidative damage to DNA, proteins, and lipids that can lead to mutations and cancer progression. This reduction in oxidative stress also disrupts the oncogenic signaling pathways that are often activated by ROS, such as the NF-κB and HIF pathways, thereby inhibiting tumor cell proliferation and survival (Jiao et al., 2016).
Furthermore, Sanghuangporus polysaccharides can induce apoptosis in tumor cells by modulating the expression of apoptosis-related genes. For instance, they have been shown to enhance the activation of p53-related genes, which are crucial for the induction of apoptosis in cancer cells (Wan et al., 2020). This activation leads to the downregulation of anti-apoptotic proteins like Bcl-2 and the upregulation of pro-apoptotic proteins like Bax, thereby promoting cell death in tumor cells. Additionally, Sanghuangporus polysaccharides can arrest the cell cycle at specific phases, such as the G0/G1 and S phases, further inhibiting tumor cell proliferation (Shao et al., 2014).
Another key mechanism by which Sanghuangporus polysaccharides exert their antitumor effects is through the regulation of the tumor microenvironment. These polysaccharides can modify the immune landscape within tumors, promoting the infiltration and activation of immune cells that target cancer cells. For instance, polysaccharide components such as SSP1 were extracted and isolated from Sanghuangporus derived from mulberry and analyzed for their structure and monosaccharide composition. Network pharmacology and molecular docking analysis revealed that SSP1 might inhibit HepG2 tumor cells through core targets such as VEGFA and AR. These polysaccharides can also enhance the activity of macrophages and T cells, which is crucial for initiating an effective antitumor immune response. This immunomodulatory effect is partially mediated by the antioxidant properties of Sanghuangporus polysaccharides, which reduce the immunosuppressive effects of oxidative stress in the tumor microenvironment (Yu et al., 2021b).
Sanghuangporus polysaccharides inhibit tumor growth through a multifaceted approach that involves reducing oxidative stress, inducing apoptosis, arresting the cell cycle, and modulating the tumor microenvironment. These combined actions make Sanghuangporus polysaccharides promising candidates for cancer therapy, particularly in strategies aimed at targeting oxidative stress and enhancing immune responses against tumors.
6 Combined Application of Sanghuangporus Polysaccharides with Conventional Antitumor Drugs
6.1 Antitumor effects of combined drug therapy
Sanghuangporus polysaccharides, as the main active components of the medicinal fungus Sanghuangporus, have been shown to effectively inhibit tumor growth and metastasis and induce apoptosis in tumor cells. Additionally, when used in combination with conventional chemotherapy drugs, Sanghuangporus polysaccharides can enhance antitumor effects. The mechanisms involved include modulating immune responses and directly acting on tumor cells, which can improve the efficacy of traditional chemotherapy, leading to better treatment outcomes and reduced side effects. For example, polysaccharides extracted from Sanghuangporus vaninii (SVP) have demonstrated the ability to inhibit tumor cell proliferation, induce apoptosis, and reduce the migration and invasion of cancer cells, particularly in breast cancer models (Wan et al., 2020). When combined with conventional chemotherapy drugs, these polysaccharides enhance antitumor effects by modulating the tumor microenvironment and boosting immune responses (Wang et al., 2022b; Li et al., 2023).
Studies have shown that natural polysaccharides, including those from Sanghuangporus, can significantly increase the efficacy of chemotherapy drugs such as cisplatin in ovarian cancer by inhibiting cell proliferation, inducing apoptosis, and enhancing immunomodulatory activities (Wang et al., 2022b). This synergistic effect not only improves the antitumor efficacy but also helps in mitigating the adverse effects commonly associated with chemotherapy, making the treatment more tolerable for patients.
6.2 Molecular mechanisms of combined drug therapy
The molecular mechanisms underlying the enhanced antitumor effects of combined therapy involving Sanghuangporus polysaccharides and conventional drugs are multifaceted. One of the key mechanisms is the activation of the p53 signaling pathway, which plays a crucial role in regulating cell cycle arrest and apoptosis. SVP, for example, has been shown to enhance the activation of p53-related genes and down-regulate matrix metalloproteinase (MMP) expression, thereby inhibiting tumor growth and metastasis (Wan et al., 2020).
Additionally, polysaccharides can modulate the tumor microenvironment by enhancing the immune response. They activate both non-specific and specific immune responses, leading to the inhibition of tumor proliferation and metastasis (Figure 7) (Ying and Hao, 2023). Polysaccharides also improve the delivery and efficacy of chemotherapy drugs through the use of stimulus-responsive nanomedicines, which allow for controlled drug release and targeted delivery to the tumor site (Li et al., 2023). This targeted approach not only maximizes the therapeutic effects but also minimizes systemic toxicity.
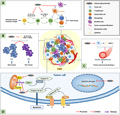 Figure 7 Anti-tumor mechanism of natural polysaccharides (Adopted from Ying and Hao, 2023) Image caption: Polysaccharides not only induce apoptosis by directly acting on tumor cells, but also inhibit the occurrence and development of tumors by acting on tumor microenvironment (TME); Among them, (A) indicates that natural polysaccharides promote the expression of cytokines such as IL-2, IFN-γ and IL-12, inhibit the expression of cytokines such as IL-4, IL-5, IL-10 and IL-13, resluting in promoting the differentiation of Th0 cells into Th1 cells, which have anti-tumor effect. What’s more, natural polysaccharides activate DC cells, allowing them to function normally in antigen presentation; (B) indicates that polysaccharide reduces the concentration of cytokines such as IL-6, IL-10, COX-2 and TGF-β in TME, so as to promote the differentiation of M1 macrophages,which play an anti-tumor role; (C) indicates that polysaccharides down regulate the expression of VEGF and so inhibit tumor related angiogenesis; (D) shows the direct anti-tumor effect of natural polysaccharides: cell cycle arrest and inducing apoptosis (Adopted from Ying and Hao, 2023) |
Chemical modifications of polysaccharides, such as sulfation, phosphorylation, and selenylation, have been shown to further enhance their antitumor activities and improve their physicochemical properties, such as water solubility and molecular weight (Xie et al., 2020; Li et al., 2021). These modifications can lead to more effective interactions with conventional chemotherapy drugs, resulting in a more robust antitumor response.
6.3 Clinical prospects of combined therapy
The clinical prospects of combining Sanghuangporus polysaccharides with conventional antitumor drugs are highly promising. The synergistic effects observed in preclinical studies suggest that such combinations could lead to more effective and less toxic cancer treatments. Polysaccharides' ability to enhance the efficacy of chemotherapy drugs while reducing their side effects makes them attractive candidates for adjuvant therapy in clinical settings (Chen and Huang, 2018; Yang et al., 2021).
Moreover, the use of polysaccharides in combination with other therapeutic modalities, such as immunotherapy, photodynamic therapy, and photothermal therapy, has shown potential in preclinical studies. These combination strategies can lead to improved antitumor efficacy by targeting multiple pathways and mechanisms involved in tumor growth and progression (Li et al., 2023).
The broad application prospects of polysaccharides are further supported by their non-toxic nature and ability to enhance the quality of life for cancer patients. As more efficient polysaccharides with antitumor activities are discovered, their integration into clinical practice is expected to grow, providing new avenues for cancer treatment (Yang et al., 2021).
The combined application of Sanghuangporus polysaccharides with conventional antitumor drugs holds significant potential for improving cancer treatment outcomes. The synergistic effects, molecular mechanisms, and clinical prospects discussed herein highlight the importance of further research and development in this area to fully realize the therapeutic benefits of such combinations.
7 Potential of Sanghuangporus Polysaccharides in Cancer Prevention
7.1 Anti-inflammatory and immunomodulatory effects of Sanghuangporus polysaccharides
Sanghuangporus polysaccharides have demonstrated significant anti-inflammatory and immunomodulatory effects, which are crucial in cancer prevention. These polysaccharides can modulate the immune system by enhancing the activity of various immune cells, such as lymphocytes and myeloid cells, thereby boosting the body's natural defense mechanisms against cancerous cells (Ren et al., 2020). The immunomodulatory activities of these polysaccharides are attributed to their ability to stimulate both innate and adaptive immune responses, which are essential for identifying and eliminating tumor cells (Ramberg et al., 2010; Ren et al., 2020).
Moreover, the anti-inflammatory properties of Sanghuangporus polysaccharides help in reducing chronic inflammation, a known risk factor for cancer development. Chronic inflammation can lead to DNA damage, promote tumor growth, and create an environment conducive to cancer progression. By mitigating inflammation, Sanghuangporus polysaccharides can potentially lower the risk of cancer initiation and progression (Wang et al., 2022b; Pradhan et al., 2023). These combined anti-inflammatory and immunomodulatory effects make Sanghuangporus polysaccharides a promising candidate for cancer prevention strategies.
7.2 Application of Sanghuangporus polysaccharides in early cancer prevention
The application of Sanghuangporus polysaccharides in early cancer prevention is supported by their ability to inhibit tumor growth and induce apoptosis in cancer cells. Studies have shown that these polysaccharides can regulate cell cycle progression, promote apoptosis, and reduce the migratory and invasive capacities of cancer cells. For instance, polysaccharides from Sanghuangporus vaninii have been found to activate the p53 signaling pathway, which plays a critical role in controlling cell proliferation and apoptosis in breast cancer cells (Wan et al., 2020). This suggests that Sanghuangporus polysaccharides can be effective in preventing the early stages of cancer by targeting and eliminating abnormal cells before they develop into malignant tumors.
Additionally, the antioxidant properties of Sanghuangporus polysaccharides contribute to their cancer-preventive effects. Antioxidants neutralize free radicals, which can cause oxidative stress and DNA damage, leading to cancer. By scavenging these free radicals, Sanghuangporus polysaccharides help protect cells from oxidative damage and reduce the likelihood of cancer development (Wang et al., 2022b; Pradhan et al., 2023). These properties highlight the potential of Sanghuangporus polysaccharides as a natural and effective means of early cancer prevention.
7.3 Application of Sanghuangporus polysaccharides in dietary supplements
The incorporation of Sanghuangporus polysaccharides into dietary supplements offers a practical approach to harness their cancer-preventive properties. As dietary supplements, these polysaccharides can be consumed regularly to provide continuous support to the immune system and maintain overall health. The safety and tolerability of polysaccharides from various sources, including Sanghuangporus, have been well-documented, making them suitable for long-term use as dietary supplements (Ramberg et al., 2010; Li et al., 2021).
Dietary polysaccharides have been shown to exert significant immunomodulatory effects in both animal and human studies, enhancing immune function and potentially improving the body's ability to fight off cancerous cells. For example, glucan extracts from mushrooms have been found to improve survival and immune function in cancer patients, suggesting that similar benefits could be achieved with Sanghuangporus polysaccharides (Ramberg et al., 2010). Furthermore, the use of these polysaccharides as dietary supplements can provide additional health benefits, such as improved gut health and reduced inflammation, which are important for overall well-being and cancer prevention (Wang et al., 2022b; Pradhan et al., 2023).
Sanghuangporus polysaccharides hold great potential in cancer prevention through their anti-inflammatory, immunomodulatory, and antioxidant properties. Their application in early cancer prevention and as dietary supplements offers a promising strategy for reducing cancer risk and promoting health. Continued research and development of Sanghuangporus polysaccharides will further elucidate their mechanisms of action and optimize their use in cancer prevention and health maintenance.
8 Future Research Directions and Challenges
8.1 In-depth research on molecular mechanisms of Sanghuangporus polysaccharides
The molecular mechanisms underlying the antitumor effects of Sanghuangporus polysaccharides remain an area ripe for exploration. Current studies have shown that these polysaccharides can regulate cell cycles, promote apoptosis, and reduce the migratory and invasive capacities of cancer cells. For instance, Sanghuangporus vaninii polysaccharides (SVP) have been found to activate p53-related genes and down-regulate matrix metalloproteinase (MMP) expression, thereby inhibiting tumor growth and metastasis in breast cancer cells (Wan et al., 2020). However, the precise molecular pathways and interactions involved are not fully understood. Future research should focus on elucidating these mechanisms through advanced molecular biology techniques such as transcriptomics, proteomics, and metabolomics. Additionally, the role of structural modifications of polysaccharides, such as sulfation and phosphorylation, in enhancing their antitumor activity should be investigated (Huang and Huang, 2017). Understanding these mechanisms will provide a solid foundation for developing more effective Sanghuangporus polysaccharide-based therapies.
8.2 Challenges in developing Sanghuangporus polysaccharides as antitumor drugs
Despite the promising antitumor properties of Sanghuangporus polysaccharides, several challenges hinder their development into clinically viable drugs. One major challenge is the variability in polysaccharide composition and bioactivity due to differences in extraction methods, sources, and strains of Sanghuangporus (Wang et al., 2022b; Wang and Huang, 2024). Standardizing the extraction and purification processes is crucial to ensure consistent quality and efficacy. Another challenge is the limited understanding of the pharmacokinetics and bioavailability of these polysaccharides in the human body. Studies have shown that polysaccharides can have significant antitumor effects in vitro and in animal models, but their effectiveness in humans remains to be validated through clinical trials (Zhao et al., 2021). Additionally, the potential side effects and toxicity of long-term use of Sanghuangporus polysaccharides need to be thoroughly assessed. Addressing these challenges requires a multidisciplinary approach involving pharmacologists, biochemists, and clinical researchers.
8.3 Clinical application prospects of Sanghuangporus polysaccharides
The clinical application of Sanghuangporus polysaccharides holds great promise, particularly in the field of oncology. Their ability to enhance immune function, as demonstrated by the increased secretion of cytokines such as IL-2, IL-6, and IFN-γ, positions them as potential adjuvants in cancer therapy (Zhu et al., 2023). Moreover, the antioxidant and anti-inflammatory properties of these polysaccharides could help mitigate the side effects of conventional cancer treatments such as chemotherapy and radiation therapy. Clinical trials are essential to validate the efficacy and safety of Sanghuangporus polysaccharides in human patients. For example, a meta-analysis of Grifola frondosa polysaccharides has shown significant tumor inhibition and immune enhancement in preclinical studies, suggesting a potential for similar outcomes with Sanghuangporus polysaccharides (Zhao et al., 2021). Furthermore, the integration of Sanghuangporus polysaccharides into functional foods and dietary supplements could provide a preventive approach to cancer, leveraging their health-promoting properties (Wang et al., 2022b). However, rigorous clinical studies and regulatory approvals are necessary to translate these prospects into reality.
While Sanghuangporus polysaccharides exhibit significant antitumor potential, further research is needed to fully understand their molecular mechanisms, overcome development challenges, and realize their clinical application. Collaborative efforts across various scientific disciplines will be key to unlocking the therapeutic potential of these natural compounds.
9 Concluding Remarks
The studies on Sanghuangporus polysaccharides have demonstrated significant antitumor effects across various cancer models. Specifically, Sanghuangporus vaninii polysaccharides (SVP) have shown to inhibit tumor proliferation, regulate the cell cycle, promote apoptosis, and reduce the migratory and invasive capacities of cancer cells, particularly in breast cancer MCF-7 cells. The mechanism involves the activation of p53-related genes and down-regulation of MMP expression. Additionally, polysaccharides from other sources, such as Grifola frondosa and Actinidia eriantha, have also exhibited potent antitumor activities by enhancing immune responses and inducing apoptosis in tumor cells. The structural characterization of these polysaccharides reveals that their antitumor efficacy is closely related to their specific molecular compositions and structural features.
The research on Sanghuangporus polysaccharides has significantly contributed to the understanding of natural polysaccharides as potential antitumor agents. These studies have provided insights into the molecular mechanisms underlying the antitumor effects of these polysaccharides, highlighting their ability to modulate key signaling pathways, such as the p53 pathway, and to enhance immuneresponses. Furthermore, the structural analysis of these polysaccharides has elucidated the relationship between their molecular structure and biological activity, paving the way for the development of more effective polysaccharide-based therapies. The findings also underscore the potential of Sanghuangporus polysaccharides as functional foods and nutraceuticals with health-promoting properties.
The promising antitumor effects of Sanghuangporus polysaccharides suggest their potential application in cancer therapy. Future research should focus on clinical trials to validate the efficacy and safety of these polysaccharides in human subjects. Additionally, the structural modification of polysaccharides could further enhance their antitumor activity, offering new avenues for the development of novel anticancer drugs. The immunomodulatory properties of these polysaccharides also indicate their potential use in combination therapies to boost the immune response in cancer patients. Moreover, the antioxidant and anti-inflammatory activities of Sanghuangporus polysaccharides highlight their potential in the prevention and treatment of other diseases, such as ulcerative colitis, thereby broadening their therapeutic applications. By advancing our understanding of the antitumor mechanisms and therapeutic potential of Sanghuangporus polysaccharides, these studies lay the groundwork for future research and development in the field of natural product-based cancer therapies.
Acknowledgments
Thank you to the anonymous peer review for providing targeted revision suggestions for the manuscript.
Funding
This research was fiinded by agrant from Changchun Sci-Tech University Youth Fund Project 2024ZD001.
Conflict of Interest Disclosure
The authors affirm that this research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Bie N., Han L., Wang Y., Wang X., and Wang C., 2020, A polysaccharide from Grifola frondosa fruit body induces HT-29 cells apoptosis by PI3K/AKT-MAPKs and NF-κB-pathway, International Journal of Biological Macromolecules, 147: 79-88.
https://doi.org/10.1016/j.ijbiomac.2020.01.062
Cao W., Li X., Wang X., Fan H., Zhang X., Hou Y., Liu S., and Mei Q., 2010, A novel polysaccharide, isolated from Angelica sinensis (Oliv.) Diels induces the apoptosis of cervical cancer HeLa cells through an intrinsic apoptotic pathway, Phytomedicine: International Journal of Phytotherapy and Phytopharmacology, 17(8-9): 598-605.
https://doi.org/10.1016/j.phymed.2009.12.014
PMid:20092988
Carneiro B., and El-Deiry W., 2020, Targeting apoptosis in cancer therapy, Nature Reviews Clinical Oncology, 17: 395-417.
https://doi.org/10.1038/s41571-020-0341-y
PMid:32203277 PMCid:PMC8211386
Chen T., 2024, Single-cell RNA sequencing reveals new insights into tumor heterogeneity, Cancer Genetics and Epigenetics, 12(1): 55-65.
https://doi.org/10.5376/cge.2024.12.0001
Cheng J.W., Song J.L., Liu Y., Lu N., Wang Y., Hu C., He L., Wei H., Lv G., Yang S., and Zhang, Z., 2020a, Conformational properties and biological activities of α-D-mannan from Sanghuangporus Sanghuang in liquid culture, International Journal of Biological Macromolecules, 164: 3568-3579.
https://doi.org/10.1016/j.ijbiomac.2020.08.112
PMid:32810532
Cheng J., Song J., Wei H., Wang Y., Huang X., Liu Y., Lu N., He L., Lv G., Ding H., Yang S., and Zhang Z., 2020b, Structural characterization and hypoglycemic activity of an intracellular polysaccharide from Sanghuangporus Sanghuang mycelia, International Journal of Biological Macromolecules, 164: 3305-3314.
https://doi.org/10.1016/j.ijbiomac.2020.08.202
PMid:32871118
Cui H., Wu S., Shang Y., Li Z., Chen M., Li F., and Wang C., 2016, Pleurotus nebrodensis polysaccharide(PN50G) evokes A549 cell apoptosis by the ROS/AMPK/PI3K/AKT/mTOR pathway to suppress tumor growth, Food & Function, 7(3): 1616-1627.
https://doi.org/10.1039/C6FO00027D
PMid:26918909
Ding H., Chen X., Chen H., Wang C., and Qian G., 2020, Effect of Sargassum fusiforme polysaccharide on apoptosis and its possible mechanism in human erythroleukemia cells, Chinese Journal of Natural Medicines, 18(10): 749-759.
https://doi.org/10.1016/S1875-5364(20)60015-2
PMid:33039054
Gao C., Cai C., Liu J., Wang Y., Chen Y., Wang L., and Tan Z., 2020, Extraction and preliminary purification of polysaccharides from Camellia oleifera Abel. seed cake using a thermoseparating aqueous two-phase system based on EOPO copolymer and deep eutectic solvents, Food Chemistry, 313: 126164.
https://doi.org/10.1016/j.foodchem.2020.126164
PMid:31935662
Ge Q., Mao J.W., Zhang A.F., and Sun B.L., 2013, Isolation, purification and structural elucidation of polysaccharide from the fruiting bodies of Phellinus baumii Pilát, Food Technology, 38(3): 168-171,175.
Huang G., and Huang H., 2017, The derivatization and antitumor mechanisms of polysaccharides, Future Medicinal Chemistry, 9(16): 1931-1938.
https://doi.org/10.4155/fmc-2017-0132
PMid:29076350
Jiao R., Liu Y., Gao H., Xiao J., and So K., 2016, The anti-oxidant and antitumor properties of plant polysaccharides, The American Journal of Chinese Medicine, 44(3): 463-88.
https://doi.org/10.1142/S0192415X16500269
PMid:27109156
Li N., Wang C., Georgiev M., Bajpai V., Tundis R., Simal-Gándara J., Lu X., Xiao J., Tang X., and Qiao X., 2021, Advances in dietary polysaccharides as anticancer agents: structure-activity relationship, Trends in Food Science and Technology, 111: 360-377.
https://doi.org/10.1016/j.tifs.2021.03.008
Li Q., Liu X., Yan C., Zhao B., Zhao Y., Yang L., Shi M., Yu H., Li X., and Luo K., 2023, Polysaccharide-based stimulus-responsive nanomedicines for combination cancer immunotherapy, Small, 19(23): e2206211.
https://doi.org/10.1002/smll.202206211
PMid:36890780
Li Y., Zhang Q., Chen X., and Zhang A., 2022, Research progress on the separation, purification, bioactivity, and product development of Polyporus umbellatus polysaccharides, Edible Fungi, 26-31, 61.
Liu Y.H, Liu C.H., Jiang H.Q., Zhou H., Li P., and Wang F., 2015, Isolation, structural characterization and neurotrophic activity of a polysaccharide from Phellinus ribis, Carbohydrate Polymers, 127: 145-151.
https://doi.org/10.1016/j.carbpol.2015.03.057
PMid:25965467
Liu J., Song J., Gao F., Chen W., Zong Y., Li J., He Z., and Du R., 2023b, Extraction, purification, and structural characterization of polysaccharides from Sanghuangporus vaninii with anti-inflammatory activity, Molecules, 28(16): 6081.
https://doi.org/10.3390/molecules28166081
PMid:37630334 PMCid:PMC10459065
Liu X., Liu X., Mao W., Guo Y., Bai N., Jin L., Shou Q., and Fu H., 2023a, Tetrastigma polysaccharide reprogramming of tumor-associated macrophages via PPARγ signaling pathway to play antitumor activity in breast cancer, Journal of Ethnopharmacology, 314: 116645.
https://doi.org/10.1016/j.jep.2023.116645
PMid:37196813
Mei Y.X., Zhu H., Hu Q.M., Liu Y., Zhao S., Peng N., and Liang Y., 2015, A novel polysaccharide from mycelia of cultured Phellinus linteus displays antitumor activity through apoptosis, Carbohydrate Polymers, 124: 90-97.
https://doi.org/10.1016/j.carbpol.2015.02.009
Pei J.J., Wang Z.B., Ma H.L., and Yan J.K., 2015, Structural features and antitumor activity of a novel polysaccharide from alkaline extract of Phellinus linteus mycelia, Carbohydrate Polymers, 115: 472-477.
https://doi.org/10.1016/j.carbpol.2014.09.017
PMid:25439921
Pradhan B., Bhuyan P., and Ki J., 2023, Immunomodulatory, antioxidant, anticancer, and pharmacokinetic activity of ulvan, a seaweed-derived sulfated polysaccharide: an updated comprehensive review, Marine Drugs, 21(5): 300.
https://doi.org/10.3390/md21050300
PMid:37233494 PMCid:PMC10223415
Ramberg J., Nelson E., and Sinnott R., 2010, Immunomodulatory dietary polysaccharides: a systematic review of the literature, Nutrition Journal, 9: 54-54.
https://doi.org/10.1186/1475-2891-9-54
PMid:21087484 PMCid:PMC2998446
Ren L., Zhang J., and Zhang T., 2020, Immunomodulatory activities of polysaccharides from ganoderma on immune effector cells, Food Chemistry, 340: 127933.
https://doi.org/10.1016/j.foodchem.2020.127933
PMid:32882476
Shao P., Chen X., and Sun P., 2014, Chemical characterization, antioxidant and antitumor activity of sulfated polysaccharide from Sargassum horneri, Carbohydrate Polymers, 105: 260-269.
https://doi.org/10.1016/j.carbpol.2014.01.073
PMid:24708979
Shu G., Jiang S., Mu J., Yu H., Duan H., and Deng X., 2018, Antitumor immunostimulatory activity of polysaccharides from Panax japonicus C. A. mey: roles of their effects on CD4+ T cells and tumor associated macrophages, International Journal of Biological Macromolecules, 111: 430-439.
https://doi.org/10.1016/j.ijbiomac.2018.01.011
PMid:29317237
Song W., Hu P., Shan Y., Du M., Liu A., and Ye R., 2014, Cartilage polysaccharide induces apoptosis in K562 cells through a reactive oxygen species-mediated caspase pathway, Food & Function, 5(10): 2486-2493.
https://doi.org/10.1039/C4FO00476K
PMid:25112602
Wan X., Jin X., Xie M., Liu J., Gontcharov A., Wang H., Lv R., Liu D., Wang Q., and Li Y., 2020, Characterization of a polysaccharide from Sanghuangporus vaninii and its antitumor regulation via activation of the p53 signaling pathway in breast cancer MCF-7 cells, International Journal of Biological Macromolecules, 163: 865-877.
https://doi.org/10.1016/j.ijbiomac.2020.06.279
PMid:32629056
Wang W., and Huang Q.K., 2024, The application prospects of artificial intelligence in molecular medicine, International Journal of Molecular Medical Science, 14(1): 16-23.
https://doi.org/10.5376/ijmms.2024.14.0003
Wang H., Ma J., Zhou M., Si J., and Cui B., 2022a, Current advances and potential trends of the polysaccharides derived from medicinal mushrooms Sanghuangporus, Frontiers in Microbiology, 13: 965934.
https://doi.org/10.3389/fmicb.2022.965934
PMid:35992671 PMCid:PMC9382022
Wang K., Cai M., Sun S., Cheng W., Zhai D., Ni Z., and Yu C., 2022b, Therapeutic prospects of polysaccharides for ovarian cancer, Frontiers in Nutrition, 9: 879111.
https://doi.org/10.3389/fnut.2022.879111
PMid:35464007 PMCid:PMC9021481
Wang K., Zhang Q., Liu Y., Wang J., Cheng Y., and Zhang Y., 2013, Structure and inducing tumor cell apoptosis activity of polysaccharides isolated from Lentinus edodes, Journal of Agricultural and Food Chemistry, 61(41): 9849-9858.
https://doi.org/10.1021/jf403291w
PMid:24063660
Wang X., Gao A., Jiao Y., Zhao Y., and Yang X., 2018, Antitumor effect and molecular mechanism of antioxidant polysaccharides from Salvia miltiorrhiza Bunge in human colorectal carcinoma LoVo cells, International Journal of Biological Macromolecules, 108: 625-634.
https://doi.org/10.1016/j.ijbiomac.2017.12.006
PMid:29233711
Wu Z., Sun H., Li J., Ma C., Zhao S., Guo Z., Lin Y., Lin Y., and Liu L., 2014, A polysaccharide from Sanguisorbae radix induces caspase-dependent apoptosis in human leukemia HL-60 cells, International journal of biological macromolecules, 70: 615-620.
https://doi.org/10.1016/j.ijbiomac.2014.06.062
PMid:25036608
Wei J., Li J.F., Wen C.B., Fang Y.X., and Ding X.H., 2016, Therapeutic effects of mulberry polysaccharide on HCC H22 tumor-bearing mice, Traditional Chinese Medicine, 39(12): 2868-2870
Xie L., Shen M., Hong Y., Ye H., Huang L., and Xie J., 2020, Chemical modifications of polysaccharides and their anti-tumor activities, Carbohydrate Polymers, 229: 115436.
https://doi.org/10.1016/j.carbpol.2019.115436
PMid:31826393
Xu H., Wu Y., Xu S., Sun H., Chen F., and Yao L., 2009, Antitumor and immunomodulatory activity of polysaccharides from the roots of Actinidia eriantha, Journal of Ethnopharmacology, 125(2): 310-317.
https://doi.org/10.1016/j.jep.2009.06.015
PMid:19559777
Xu J., Tan Z., Shen Z., Shen X., and Tang S., 2021, Cordyceps cicadae polysaccharides inhibit human cervical cancer hela cells proliferation via apoptosis and cell cycle arrest, Food and Chemical Toxicology, 148: 111971.
https://doi.org/10.1016/j.fct.2021.111971
PMid:33421460
Yan J.K., Wang Y.Y,. Ma H.L., Wang Z.B., and Pei J.J., 2016a, Structural characteristics and antioxidant activity in vivo of a polysaccharide isolated from Phellinus linteus mycelia, Journal of the Taiwan Institute of Chemical Engineers, 65: 110-117.
https://doi.org/10.1016/j.jtice.2016.05.052
Yan J.K., Wang Y.Y., Wang Z.B., Ma H.L., Pei J.J., and Wu J.Y., 2016b, Structure and antioxidative property of a polysaccharide from an ammonium oxalate extract of Phellinus linteus, International Journal of Biological Macromolecules, 91: 92-99.
https://doi.org/10.1016/j.ijbiomac.2016.05.063
PMid:27212213
Yang S., Chen X., Sun J., Qu C., and Chen X., 2021, Polysaccharides from traditional Asian food source and their antitumor activity, Journal of Food Biochemistry, 46(3): e13927.
https://doi.org/10.1111/jfbc.13927
Yin J., Jiang Z., Yu H., Xie M., Hsiao W., Lu A., and Han Q., 2012, A new application of an aqueous diphase solvent system in one-step preparation of polysaccharide from the crude water extract of Radix Astragali by high-speed counter-current chromatography, Journal of Chromatography A, 1262: 92-97.
https://doi.org/10.1016/j.chroma.2012.08.099
Mid:22999203
Ying J., 2023, Extraction, Purification, and in vitro Antitumor Activity of Polysaccharides from Mulberry-Cultivated Sanghuangporus (Morus alba L.), Thesis for M.S., Biology and Medicine, Hebei University of Economics and Busines, Supervisor: Liu K., and Chen H., pp.1-19.
https://doi.org/10.27106/d.cnki.ghbju.2023.000802
Ying R.F., Wu C.E., Huang M.G., and Wang Y.S., 2017, Anti-tumor activity of polysaccharides from Phellinus igniarius fruiting body and mycelium, China Food Additives, 24(12): 57-61.
https://doi.org/10.ssss/j.issn.1006-2513.2017.12.006
Ying Y., and Hao W., 2023, Immunomodulatory function and anti-tumor mechanism of natural polysaccharides: a review, Frontiers in Immunology, 14: 1147641.
https://doi.org/10.3389/fimmu.2023.1147641
PMid:36969152 PMCid:PMC10035574
Yu J., Dong X., Jiao J., Ji H., and Liu A., 2021, Antitumor and immunoregulatory activities of a novel polysaccharide from Astragalus membranaceus on S180 tumor-bearing mice, International Journal of Biological Macromolecules, 189: 930-938.
https://doi.org/10.1016/j.ijbiomac.2021.08.099
PMid:34419546
Yu S., Dong X., Ji H., Yu J., and Liu A., 2021a, Antitumor activity and immunomodulation mechanism of a novel polysaccharide extracted from Polygala tenuifolia Willd. evaluated by S180 cells and S180 tumor-bearing mice, International Journal of Biological Macromolecules, 192: 546-556.
https://doi.org/10.1016/j.ijbiomac.2021.10.025
PMid:34648800
Yu S., Ji H., Dong X., Liu A., and Yu J., 2020b, FAS/FAS-L-mediated apoptosis and autophagy of SPC-A-1 cells induced by water-soluble polysaccharide from Polygala tenuifolia, International Journal of Biological Macromolecules, 150: 449-458.
https://doi.org/10.1016/j.ijbiomac.2020.02.010
PMid:32027895
Yuan Q., Zhao L.Y., Li Z.H., Harqin C., Peng Y., and Liu J., 2018, Physicochemical analysis, structural elucidation and bioactivities of a high-molecular-weight polysaccharide from Phellinus igniarius mycelia, International Journal of Biological Macromolecules, 120: 1855-1864.
https://doi.org/10.1016/j.ijbiomac.2018.09.192
Zhang S., Nie S., Huang D., Feng Y., and Xie M., 2014a, A novel polysaccharide from Ganoderma atrum exerts antitumor activity by activating mitochondria-mediated apoptotic pathway and boosting the immune system, Journal of Agricultural and Food Chemistry, 62(7): 1581-1589.
https://doi.org/10.1021/jf4053012
PMid:24506418
Zhang S., Nie S., Huang D., Huang J., Feng Y., and Xie M., 2014b, Ganoderma atrum polysaccharide evokes antitumor activity via cAMP-PKA mediated apoptotic pathway and down-regulation of Ca(2+)/PKC signal pathway, Food and Chemical Toxicology, 68: 239-246.
https://doi.org/10.1016/j.fct.2014.03.020
PMid:24704042
Zhang Z.F., Lv G.Y., Song T.T., Jin Q.L., Huang J.B., Fan L.F., and Cai W.M., 2015, Comparison of the preliminary characterizations and antioxidantproperties of polysaccharides obtained from Phellinus baumii growth on differentculture substrates, Carbohyd, Polym, 132: 397-399.
https://doi.org/10.1016/j.carbpol.2015.06.006
PMid:26256363
Zhang Z., Song T., Chen J., and Lv G., 2023, Recovery of a hypolipidemic polysaccharide from artificially cultivated Sanghuangporusporus vaninii with an effective method., Frontiers in Nutrition, 9: 1095556.
https://doi.org/10.3389/fnut.2022.1095556
PMid:36712537 PMCid:PMC9880258
Zhao F., Guo Z., Ma Z., Ma L., and Zhao J., 2021, Antitumor activities of grifola frondosa (maitake) polysaccharide: a meta-analysis based on preclinical evidence and quality assessment, Journal of Ethnopharmacology, 280: 114395.
https://doi.org/10.1016/j.jep.2021.114395
PMid:34271115
Zhou L., Fu Y., Zhang X., Wang T., Wang G., Zhou L., Yu H., and Tian X., 2023, Transcriptome and metabolome integration reveals the impact of fungal elicitors on triterpene accumulation in Sanghuangporus Sanghuang, Journal of Fungi, 9(6): 604.
https://doi.org/10.3390/jof9060604
PMid:37367540 PMCid:PMC10299497
Zhu X., Guo R., Su X., Shang K., Tan C., Ma J., Zhang Y., Lin D., Ma Y., Zhou M., Yang J., Wu Q., Sun J., Wang Z., Guo Y., Su R., Cui X., Han J., Lü Y., and Yue C., 2023, Immune-enhancing activity of polysaccharides and flavonoids derived from Phellinus igniarius YASH1, Frontiers in Pharmacology, 14: 1124607.
https://doi.org/10.3389/fphar.2023.1124607
PMid:37180713 PMCid:PMC10166811
(1).png)
. HTML
Associated material
. Readers' comments
Other articles by authors
. Haiyan Chen
. Pengfei Hu
. Li Liang
. Ke Li
. Binghui Liu
. Liujing Zhou
. Xinci Zheng
Related articles
. Sanghuangporus polysaccharides
. Antitumor effects
. Apoptosis
. Oxidative stress
. Cancer therapy
Tools
. Post a comment